Tezacaftor
CFTR corrector
General information
N/A
Synonyms
VX-661
Marketed as
Dietary sources
N/A
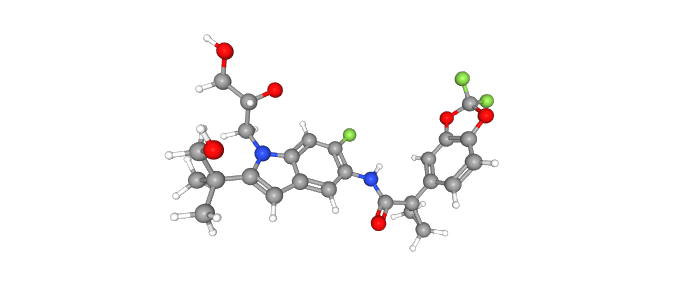
C26H27F3N2O6
Drug-Mutation Relation
results for D1152H / F508del
See all data on TezacaftorTreats
Does not treat
| Mutation | Link | Tested on | Impact factor | Notes |
|---|
