Tezacaftor
CFTR corrector
General information
N/A
Synonyms
VX-661
Marketed as
Dietary sources
N/A
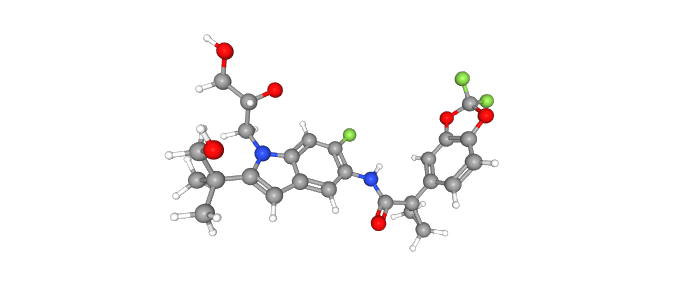
C26H27F3N2O6
Drug-Mutation Relation
Treats
Does not treat
| Mutation | Number of sources | Average impact factor | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
F508del/4016insT
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/1717-1G->A
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/S341P6
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/2183AA->G
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/2183delAA->G
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/Q290X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/W1089X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/L1065P
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/3850-3T->G
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/1677delTA
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/A46D6
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/C276X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/E585X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/S1255X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/Q493X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/Q525X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/3876delA
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/T338I8
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/3905insT
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/K710X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/G85E
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/N1303K
|
2 | 4.12 | ||||||||||||
|
||||||||||||||
|
||||||||||||||
|
F508del/V520F
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/1154insTC
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/1811+1.6kbA->G
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/574delA
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/1341+1G->A
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/541delC
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/L732X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/R785X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/Q220X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/3007delG
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/R709X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/2347delG
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/3120+1G->A
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/Y569D6
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/2143delT
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/3737delA
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/2184delA
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/3791delC
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/S466X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/3659delC
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/405+1G->A
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/Y122X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/CFTRdel2,3
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/E822X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/S1196X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/R1066C
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/R764X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/I507del
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/G542X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/R553X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/R1158X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/G330X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/R560S
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/R347P
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/2307insA
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/E92X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/A561E
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/1078delT
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/E60X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/A559T6
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/4040delA
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/W57X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/711+5G->A
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/3120G->A
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/L218X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/Q98X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/E1104X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/3600+2insT
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/2184insA
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/H1085R6
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/W1204X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/711+1G->T
|
2 | 4.12 | ||||||||||||
|
||||||||||||||
|
||||||||||||||
|
F508del/1812-1G->A
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/406-1G->A
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/2711delT
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/G673X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/1248+1G->A
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/Q414X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/R1162X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/S489X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
Q1412X
|
1 | 5.22 | ||||||||||||
|
||||||||||||||
|
F508del/R560T
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/Q1313X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/4005+1G->A
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/712-1G->T
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/1898+1G->A
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/394delTT
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/Q39X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/457TAT->G
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/663delT
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/R792X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/4374+1G->T
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/W496X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/R851X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/W1282X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/L1077P6
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/S434X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/405+3A->C
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/4382delA7
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/2622+1G->A
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/2869insG
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/1811+1G->C
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/3850-1G->A
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/R75X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/R1066M
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/L927P
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/M1101K
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
Q1390X
|
1 | 5.22 | ||||||||||||
|
||||||||||||||
|
F508del/621+1G->T
|
2 | 0.00 | ||||||||||||
|
||||||||||||||
|
||||||||||||||
|
F508del/Q552X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/2043delG
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/S912X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/W846X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/W401X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/Y1092X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/Q890X
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/CFTRdele22-23
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
|
F508del/L467P6
|
1 | 0.00 | ||||||||||||
|
||||||||||||||
