Ataluren
Suppresses premature stop mutation
General information
Ataluren is a pharmaceutical drug. It was approved in the European Union in 2014 for the treatment of Duchenne muscular dystrophy and it is sold under the name Translarna™. It does not yet have approval from the FDA. Ataluren targets nonsense mutations, where it is able to skip and produce full-length protein. In relation to cystic fibrosis, our AIM tool found the data that ataluren can be beneficial for patients with nonsense mutations who are not treated with tobramycin.
Ataluren on PubChem
Ataluren on DrugBank
Ataluren on Wikipedia
Synonyms
PTC124
Marketed as
Dietary sources
N/A
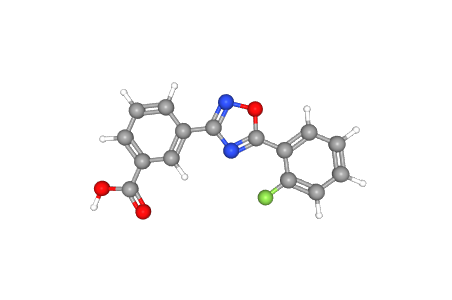
C15H9FN2O3
Drug-Mutation Relation
results for R352Q / G542X
See all data on AtalurenTreats
| Mutation | Link | Tested on | Impact factor | Notes |
|---|---|---|---|---|
| G542X | A randomized placebo-controlled trial of ataluren for the treatment of nonsense mutation cystic fibrosis | humans | 3.48 | Effective only in patients not using tobramycin. |
Does not treat
| Mutation | Link | Tested on | Impact factor | Notes |
|---|
