Amikacin
Suppresses premature stop mutation
General information
Amikacin is a small-molecule aminoglycoside antibiotic effective against resistant gram-negative bacteria, such as Pseudomonas aeruginosa and Acinetobacter baumanii. It was approved in the USA in 2018 and is sold under the brand name Arikayce®. It is used to treat Mycobacterium avium complex lung disease. In relation to cystic fibrosis, our AIM tool found the data that amikacin reduces Pseudomonas aeruginosa density, which leads to improved lung function. Amikacin can also suppress premature stop mutation (nonsense mutation) in the CFTR gene.
Amikacin on PubChem
Amikacin on DrugBank
Amikacin on Wikipedia
Synonyms
amikacin sulfate
Marketed as
Dietary sources
N/A
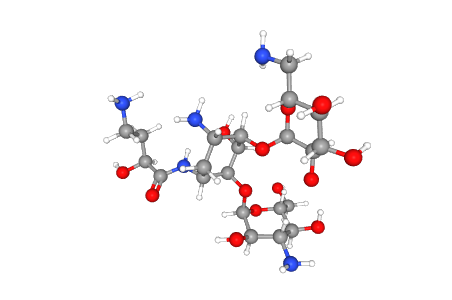
C22H43N5O13
Drug-Mutation Relation
results for W1089X / D110H
See all data on AmikacinTreats
| Mutation | Link | Tested on | Impact factor | Notes |
|---|---|---|---|---|
| General effect | Amikacin for Inhalation (Transave) (Insmed TR02-106) | humans | N/A | |
| General effect | Arikace compared to TOBI® in people with CF with chronic Pseudomonas aeruginosa infections (Insmed TR02-108) | humans | N/A | Less effective than tobramycin. |
| General effect | Amikacin for Inhalation (Transave) (Insmed TR02-106) | humans | N/A |
Does not treat
| Mutation | Link | Tested on | Impact factor | Notes |
|---|
